Selecting the right Clinical IT vendor
When embarking on the journey of selecting a new vendor to replace or to introduce new capabilities, it can be overwhelming due to the sheer number of possible solutions.
Increased complexity in clinical trials requires flexible, scalable, and easy-to-use software solutions and it is, therefore, important to consider some key questions before you enter the process of selecting a new Clinical IT vendor:
- Are process and evaluation criteria well-defined?
- How do we ensure that the vendors solution match our needs and processes?
- Will we aim for best-in-breed or unified type of platform?
- How do we approach the negotiation with the vendor?
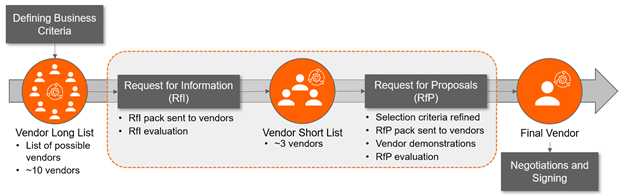
It is essential that you have clearly defined what you expect to see from a potential vendor to support your processes and needs.
Understanding your challenges and goals helps vendors offer solutions tailored to your needs, enhancing the clarity of their approach. The deliverables listed below are examples of key documents to ensure assessment of vendors in a standardized way.
- User requirement specifications (describing your requirements to the solution)
- Use Cases (describing the high-level process steps)
- Pain points and expected benefits
- Vendor Questionnaire (assessment of vendor)
This will help narrow down the list of potential vendors and reduce the time spent on evaluating those not the right fit for your organization. Additionally, this work will establish a strong foundation for the ongoing selection process and, ultimately, negotiation after selecting a vendor.
In parallel to identifying the vendor offering needed capabilities, assess the vendor’s quality level and organizational maturity to ensure alignment with your organization. Pharmaceutical companies have high-quality requirements which need to be addressed independently of how impressive a solution is. A structured approach is needed to ensure that all factors are considered.
Once the right vendor has been selected you need to consider how to approach the negotiation
For successful vendor negotiations, clarity on your organization’s priorities is crucial to achieve the best possible outcome.
By utilizing your findings during the selection process and setting key goals for what you want to achieve, you will have a much better starting point when embarking on the negotiation process.
In essence, you need to put in place a strategy for negotiating with the vendor that ensures a single, internally aligned communication stream.
A key factor in negotiating is understanding the price drivers for the selected vendor. Here it is important to have the right information available internally to fully understand which parameters affect the vendor pricing. Below are a few examples of price drivers:
- Number of studies
- Length of studies
- Number of users
When the answers to such questions are known, both in the short term (1-2 years) and the longer term (3-5 years) it allows for maximizing the positive outcome for your organization while retaining a good, long-lasting, relationship with the vendor. We can help ensure that you position yourself in the best possible way during the negotiation to get the vendor to commit to what you need for the best price.
Do you want to hear more about how we can help you on your vendor selection journey?
At HERAX, we have extensive experience with selecting the right vendor at the right price without compromising any essential capabilities.
We know the market and have experience with a vast majority of IT vendors used in the Pharmaceutical and Medical Device industry. We bring value to our clients with a combination of competencies in life science operational processes, information technology, and pharma architecture methodology.
Curious to know more? Read other articles here.

John Aggerholm
CEO & Project Director
John Aggerholm is the founder of the company. Before founding HERAX, John worked for 7 years for a daughter company Novo Nordisk A/S, as Vice President for eClinical Process Consulting, specializing in the improvement of clinical processes in R&D and the design and execution of Digitalization and eClinical programs. Before joining the Novo group, John worked 5 years as Chief Information Officer at IBM Denmark, and before that worked in IT and Clinical Operations at Henri Beaufour Institute Inc. in Washington D.C. (US).
